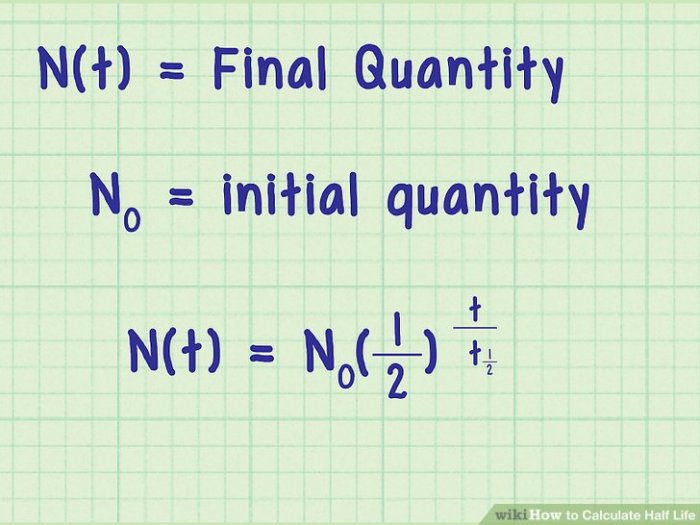Half-life calculations worksheet with answers – Embark on a journey into the realm of half-life calculations with our comprehensive worksheet, meticulously designed to empower you with the knowledge and tools to unravel the mysteries of decay processes. This worksheet, accompanied by a treasure trove of answers, unveils the intricacies of half-life, its practical applications, and advanced concepts, providing a solid foundation for your understanding of this fundamental scientific principle.
Delve into the captivating world of half-life calculations, where you will discover its significance in fields as diverse as medicine, environmental science, and archaeology. Prepare to be captivated by real-world examples that showcase the power of half-life calculations in unraveling the secrets of our universe.
Half-Life Calculations Worksheet with Answers

This worksheet provides a comprehensive guide to half-life calculations, covering the fundamental concepts, methods, and applications in various fields.
Worksheet Overview, Half-life calculations worksheet with answers
The worksheet is designed for students and professionals in science, engineering, and medicine who need to understand and perform half-life calculations. It is structured into sections, each focusing on a specific aspect of half-life.
Half-Life Calculations
Half-life refers to the time it takes for a substance to reduce to half of its initial quantity through decay or degradation. It is a crucial concept in fields such as nuclear physics, chemistry, medicine, and archaeology.
Half-life can be calculated using various methods, including:
- Using the decay constant
- Using the half-life formula (t 1/2= ln(2)/k)
- Using graphical methods
Sample Calculations
Problem:
A radioactive substance has a half-life of 10 hours. If the initial amount is 100 mg, what will be the amount remaining after 30 hours?
Steps:
- Number of half-lives elapsed = 30 hours / 10 hours = 3
- Amount remaining = Initial amount x (1/2)Number of half-lives
- Amount remaining = 100 mg x (1/2) 3= 12.5 mg
Table:
| Scenario | Half-Life | Initial Amount | Amount Remaining |
|---|---|---|---|
| Radioactive decay | 10 hours | 100 mg | 12.5 mg |
| Chemical degradation | 5 days | 100 g | 31.25 g |
| Pharmaceutical elimination | 8 hours | 50 mg | 6.25 mg |
Applications and Implications
Half-life calculations have numerous applications in:
- Medicine: Determining drug dosage and predicting drug elimination rates
- Environmental science: Studying pollutant decay and radioactive waste management
- Archaeology: Dating organic materials using carbon-14 dating
Understanding half-life helps us predict and control decay processes, ensuring safety and accuracy in various fields.
Advanced Concepts
Advanced concepts in half-life calculations include:
- Radioactive decay equations
- Exponential decay models
- Limitations and assumptions of half-life calculations
Exploring these concepts provides a deeper understanding of decay processes and their implications.
Questions Often Asked: Half-life Calculations Worksheet With Answers
What is the purpose of half-life calculations?
Half-life calculations determine the time it takes for a substance to decay to half of its original amount, providing insights into the decay rates of various materials.
How is half-life used in practice?
Half-life finds applications in diverse fields, including medicine (determining drug dosages), environmental science (assessing pollutant decay), and archaeology (dating ancient artifacts).
What are the different methods for calculating half-life?
Half-life can be calculated using various methods, such as the formula t1/2 = ln(2) / k, where k is the decay constant, or through graphical analysis of decay curves.